top of page
Digital Health
FDA-AUTHORIZED THERAPY SOLUTIONS FOR PATIENTS AND PROVIDERS
Company
Pear Therapeutics, 2019-2021
Challenge
Pear created FDA-authorized patient apps and provider portals to manage treatment. This meant we needed to show statistically significant positive outcomes for patients, which we did. However, the company's biggest moat (FDA authorization) was also the hardest challenge: patients need a prescription to get the app.
My Role
I was a product manager focused on evidence-based health content creation and design, both the patient and provider experience. I was hired in part because of my games background, since gamified elements greatly drove engagement. The key parts of my role included: advocating for product strategy, managing a roadmap, project managing features and clinical trials, and hands-on design work focused on content design, onboarding, and gamification.
Results
My products, Reset (for substance use disorder) and Somryst (for insomnia) greatly contributed to an IPO in 2021 at a $1.6 billion valuation

Somryst for Insomnia
Insomnia-specific cognitive behavioral therapy. Worked with SMEs (subject matter experts) on the best content and design to support patients through sleep restriction, which is actually a key component to improved sleep. Because of the brain fog of the patient population, we pain close attention using plain language and easy-to-navigate interactions. Created provider web experience to give robust data summary of how patients were doing, and to let providers contribute to that data journey from their own sessions or communications with patients. Partnered with the Columbia School of Social work on a clinical trial.
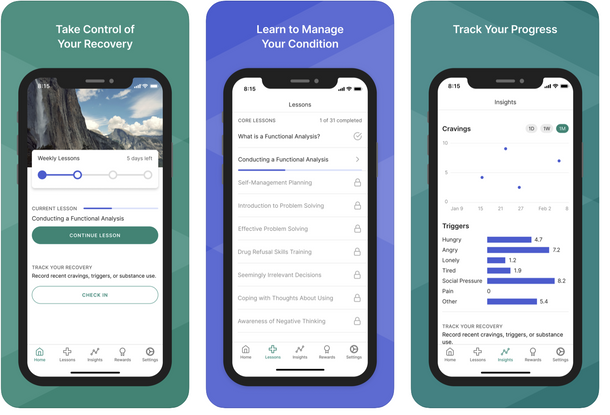

Reset for Substance Use
Reset has both a patient app and provider web experience, and is meant to be used in tandem with in-person therapy, drug screening, and with medications such as buprenorphine for opioid use disorder or others. The app provides gamified progress through cognitive behavioral therapy and other education modules, providing a randomized gift card rewards at key moments, mirroring how traditional substance use recovery programs often work. The app has been shown to have a statistically significant lift in program engagement.



bottom of page



